 In this model n = ∞ corresponds to the level where the energy holding the electron and the nucleus together is zero. Rutherford had made the startling discovery that most of the atom is empty space. The K-shell can hold a maximum of 2 electrons, the L-shell can hold a maximum of 8 electrons, and. Bohr, one of the pioneers of quantum theory, had taken the atomic model presented a few years earlier by physicist Ernest Rutherford and given it a quantum twist. For K-shell n1, for L-shell n2, for M-shell n3, and so on. 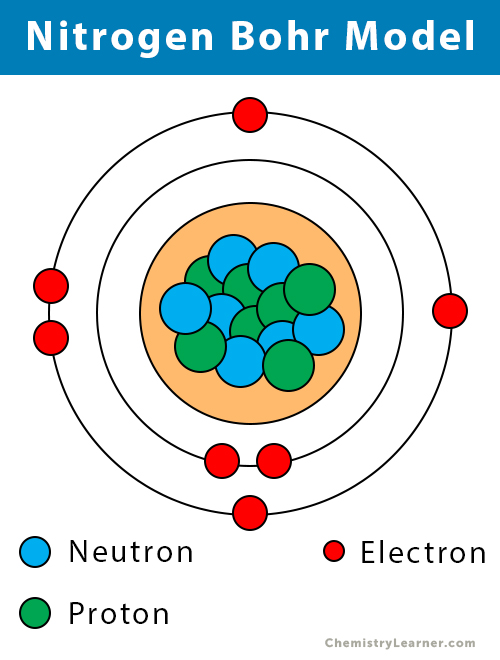
The electrons are embedded in this sphere so as to give the most stable electrostatic arrangement. He proposed that an atom is shaped like a sphere with a radius of approximately 10-10 m, where the positive charge is uniformly distributed.  
Note- ‘n’ is the number of the orbit or shell. Thomson proposed the first of many atomic models to come. \) is the Rydberg constant in terms of energy, Z is the atom is the atomic number, and n is a positive integer corresponding to the number assigned to the orbit, with n = 1 corresponding to the orbit closest to the nucleus. According to Bohr’s Atomic model, the number of electrons that an orbit or shell can hold can be calculated using the formula: 2n2. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
